

TOP

Antibody Drugs
Antibody Drugs
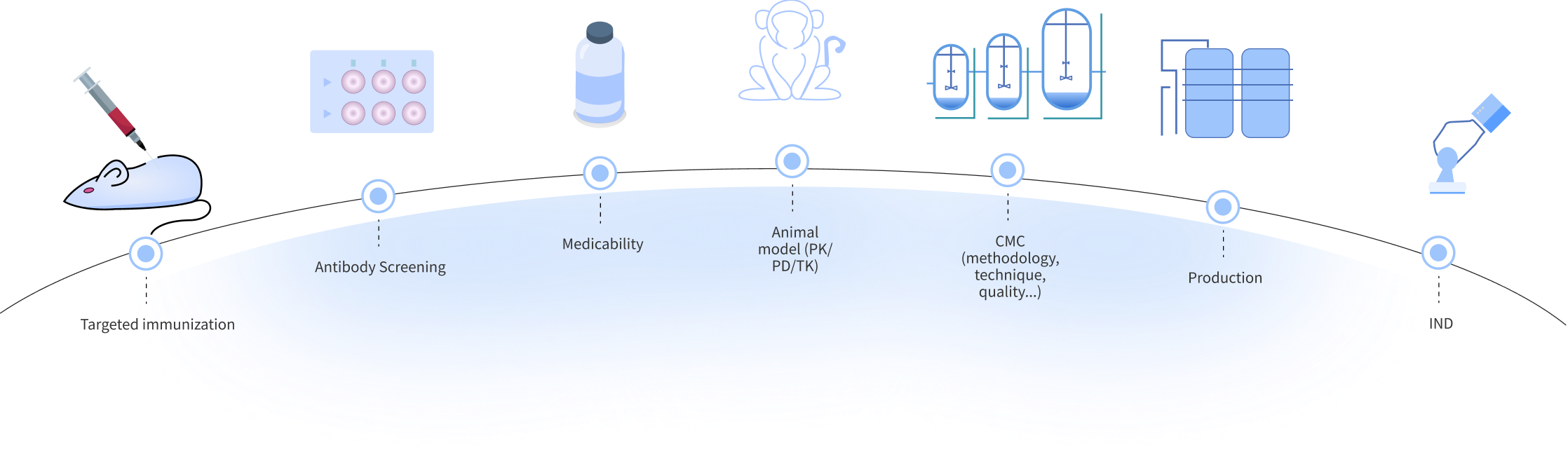
Antibody solutions include precise target identification and rigorous validation, and optimized antibody design to enhance performance. In production, we need to ensure efficient cell culture, strict purification and precise quality control. In the clinical stage, we need to scientifically design trials, accelerate IND and advance clinical trials to achieve the goal of safe and effective antibody drug development.

Target discovery and validation
In antibody drug development, disease biology research is conducted to gain an in-depth understanding of disease mechanisms, pathological processes and related molecular pathways. Then, target screening is conducted through genomics, proteomics and other technical means to identify potential targets that are closely related to the disease. Then, the screened targets are functionally verified using cell models and animal models to confirm the validity and specificity of the targets.

Antibody Design and Screening
We design suitable antibody structures, such as monoclonal antibodies, bispecific antibodies, etc., according to the characteristics of the target. Subsequently, a large-capacity antibody library is constructed by genetic engineering technology, and then antibodies with high affinity and specificity are screened and optimized from the antibody library by high-throughput screening technology.
· siRNA/miRNA/ASO synthesis service
· NGS Safety Evaluation of Protein Drugs
· CRISOPR-sgRNA library construction

Preclinical studies
In the preclinical research stage of antibody drug development, pharmacodynamic studies are conducted to evaluate the therapeutic effects of antibodies in cellular and animal models; pharmacokinetic studies are conducted to gain an in-depth understanding of the absorption, distribution, metabolism, and excretion processes of antibodies in the body; and safety evaluations are conducted, covering acute toxicity, chronic toxicity, genotoxicity, and other aspects of the study.
· NGS Safety Evaluation of Protein Drugs
If you have any questions, please contact us
