

TOP

Cell Therapy Solutions
Cell Therapy Solutions
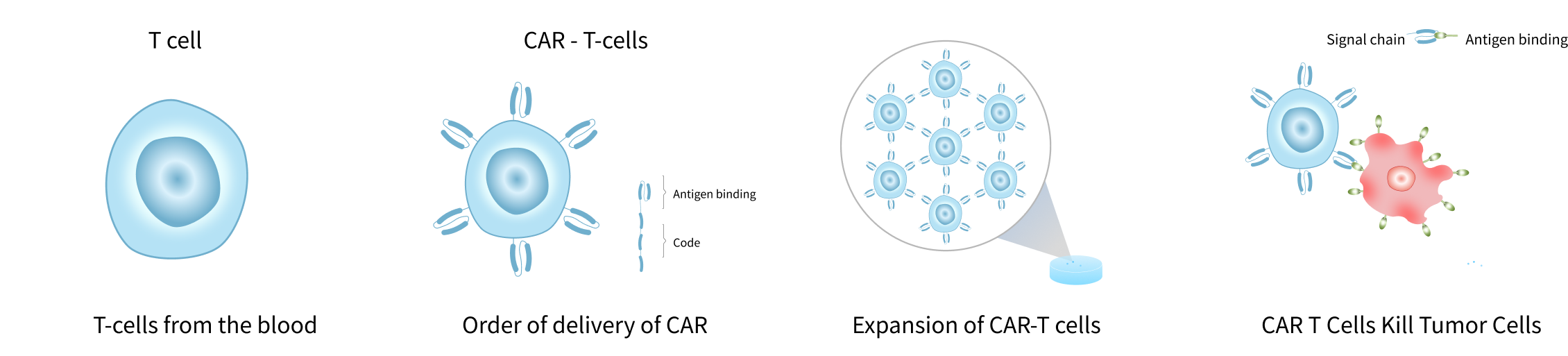
Cell therapy includes immune cell therapy and stem cell therapy, in which the patient's own cells or cells provided by the donor are cultured in vitro to achieve expansion, activation, or gene editing, etc., and then infused back into the patient's body to stimulate and strengthen the body's immune function, and ultimately to achieve control of the disease. Depending on the mechanism of action, representative cell therapies include CAR-T, TILs, TCR-T, CAR-NK, etc. GentieGen has a wide range of products and services that cover the entire process of cell therapy development. GentieGen provides high-quality and reliable services, instruments and reagents to accelerate the pace of your cellular drug from target discovery to clinical research to commercialization.
Drug Development Process

Target discovery and validation
Select targets with clear biological functions and disease relevance. For example, for tumor cell therapy, select targets that are highly expressed on tumor cells but low or absent on normal cells to improve the specificity and effectiveness of treatment.
· siRNA/miRNA/ASO synthesis service
· Eukaryotic/prokaryotic transcriptome sequencing

Cell Selection and Acquisition
The appropriate cell type is determined according to the type of disease, therapeutic mechanism and safety. For example, for immunomodulation-related diseases, regulatory T cells may be selected; for tumor therapy, T cells and K cells are often selected. Ensure the safety of cell collection and the quality and purity of cells. The collection method should be minimally damaging to the patient, and the isolation technique should be efficient and retain the activity and function of the cells.

Cell Culture and Expansion
Ensure the safety of cell collection and the quality and purity of cells. The collection method should be minimally damaging to the patient, and the isolation technique should be highly effective in preserving the activity and function of the cells.

Cellular transformation and modification
Selection of appropriate gene editing technologies or vectors ensures the accuracy and stability of the modifications while avoiding the introduction of unwanted mutations or side effects.
· Compliance Gene Synthesis Services

Quality Inspection and Control
Establish strict standards for quality control testing, including cell activity, purity, phenotype, accuracy of gene modification, sterility, and many other aspects.
· NGS Safety Evaluation of Nucleic Acid Drugs

Preclinical studies
Select appropriate animal models, fully evaluate the safety and efficacy of drugs, and determine the initial therapeutic dose and protocol. Design scientifically sound clinical trial protocols, including patient enrollment criteria, incremental therapeutic doses, and efficacy and safety assessment indicators.
· NGS Safety Evaluation of Nucleic Acid Drugs
· Safety Evaluation of Cell Therapy for NGS
· Gene Editing NGS Safety Evaluation
If you have any questions, please contact us
